Why Most PV Teams Are Not Pharmacovigilance Inspection-Ready
Most PV professionals are trained to execute tasks, not defend pharmacovigilance systems during inspection. Here is where training fails regulatory expectations.

Most pharmacovigilance teams are trained.
But very few are inspection-ready.
That gap is not about knowledge. It is about how the system performs under scrutiny.
Teams can process cases, follow SOPs, and meet timelines. Yet during inspections, uncertainty appears. Not because the work is incorrect, but because the system behind the work cannot be clearly explained, traced, or defended.
That is where inspections begin to fail.
Pharmacovigilance Inspection Readiness: What Regulators Actually Assess
Inspection expectations have shifted.
Regulators are no longer verifying whether activities are completed. They are assessing whether the pharmacovigilance system is controlled, consistent, and explainable.
This includes:
- Traceability across systems
- Alignment between SOPs and actual practice
- Visibility of QPPV oversight
- Real-time explanation of decisions
If evidence cannot be retrieved quickly or decisions cannot be justified, confidence in the system drops immediately.
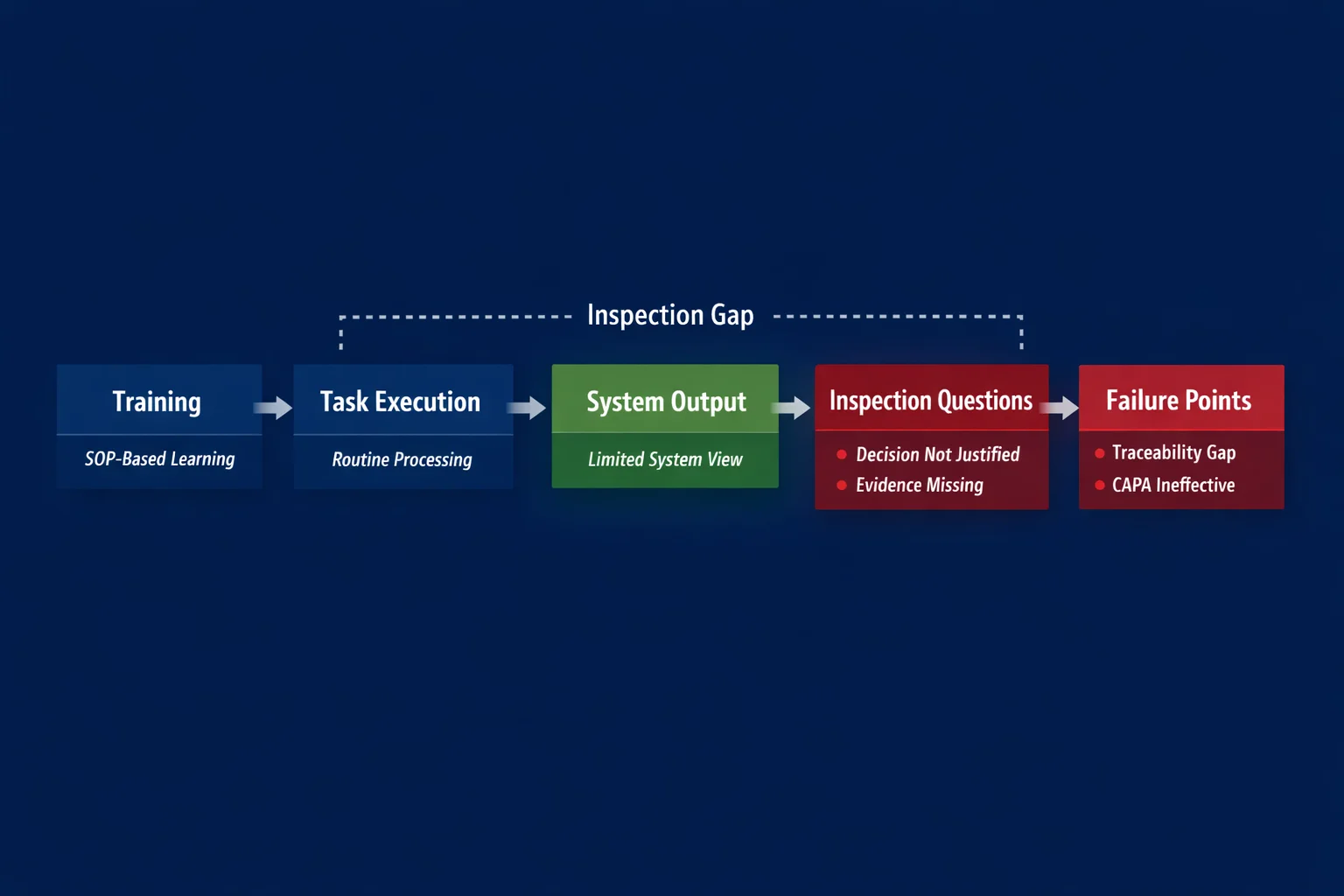
Why PV Training Does Not Translate to Inspection Readiness
Most PV training programs are built for execution. They focus on case processing accuracy, timelines and compliance, system navigation, and SOP adherence. These are essential for day-to-day operations, but they are not sufficient for inspection readiness.
Training prepares professionals to work within defined workflows, but inspections require them to step outside those workflows and explain system behavior, decisions, and risks.
That is the disconnect.
The Core Gap: Task Execution vs System Defensibility
A trained professional knows how to follow a process.
An inspection-ready professional knows how to defend it.
Inspectors ask questions that go beyond execution:
- Why was this case classified this way?
- What triggered this escalation?
- How do you know this CAPA resolved the issue?
- Where is the evidence of QPPV oversight?
Those questions expose whether the system is understood or just followed.
Where Pharmacovigilance Systems Fail During Inspections
Failures are rarely isolated. They appear across connected areas of the system.
Example: A deviation leads to retraining. The same deviation reappears. The CAPA looks complete, but effectiveness was never demonstrated. For inspectors, that is a system failure, not an isolated issue.
QPPV Oversight and Pharmacovigilance Inspection Readiness
This is where most systems break.
Regulators expect the QPPV to demonstrate:
- Continuous oversight of the PV system
- Awareness of key risks and decisions
- Involvement in signal management, CAPA, and vendor governance
In reality, many systems show:
- Oversight through dashboards, not decisions
- Limited documented involvement
- Gaps between responsibility and visibility
Example: A QPPV reviews periodic summaries but cannot show evidence of involvement in a signal escalation or vendor performance review. That creates immediate doubt about system control.
Delegation is allowed. Loss of visibility is not.
The role of Local QPPVs is also evolving. They are becoming critical in maintaining market-specific compliance and preventing delays or regulatory action at a regional level.
The Business Risk Behind Poor PV Inspection Readiness
This gap is not just regulatory. It directly affects business outcomes.
- Inspection findings delay approvals
- Weak systems increase regulatory exposure
- Safety failures can lead to recalls and litigation
Industry estimates have shown that safety and compliance failures can result in multi-billion-dollar losses globally. For high-value products, even short delays impact revenue significantly.
Inspection readiness is operational risk management.
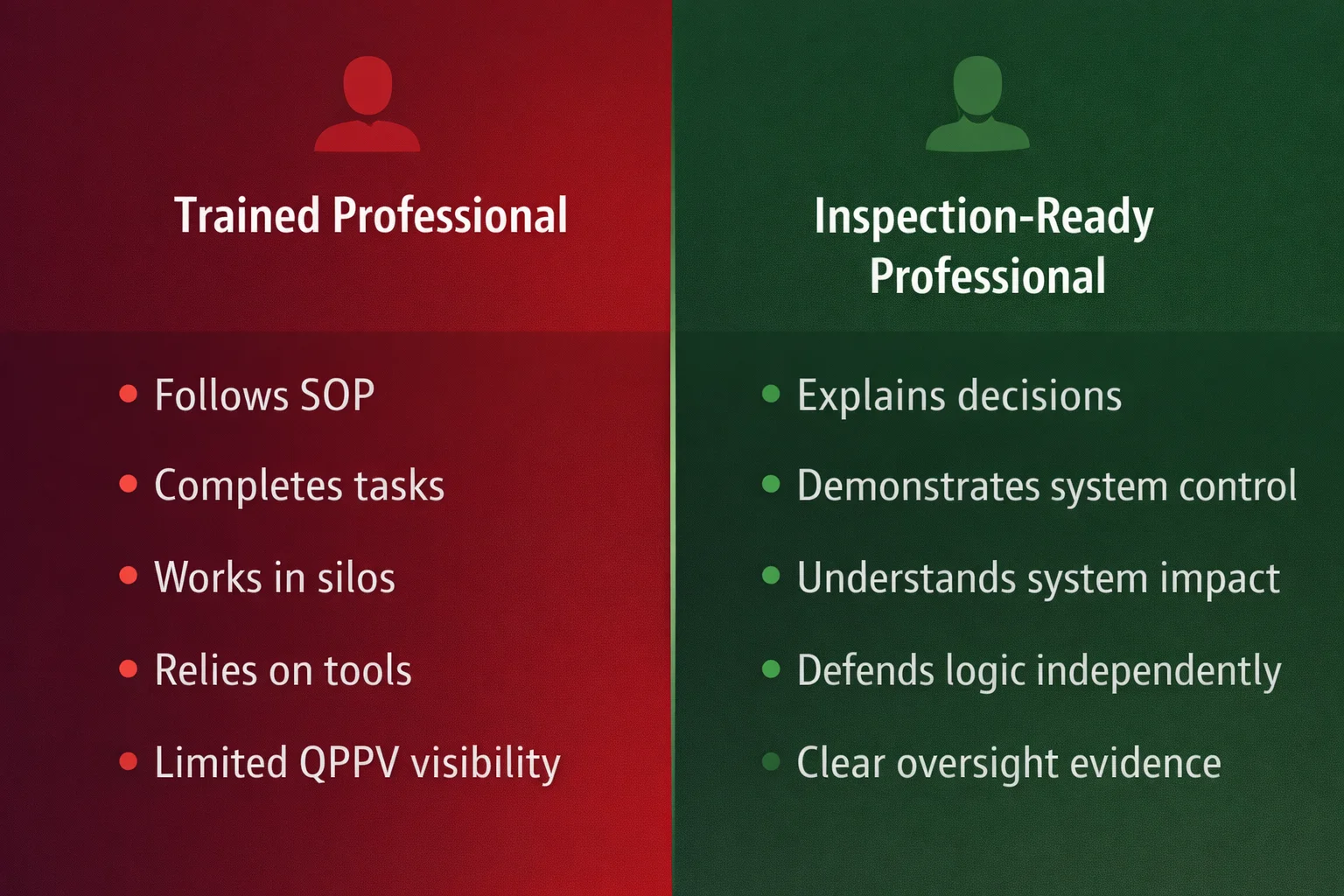
The Shift Required for Pharmacovigilance Inspection Readiness
Most teams operate in task mode. Inspection-ready teams operate in system mode.
Task-focused approach:
- Follow SOPs
- Complete activities
- Close records
System-focused approach:
- Understand decision impact
- Ensure traceability
- Validate consistency
- Demonstrate control
This shift is what inspections are actually testing.
Pharmacovigilance Inspection-Readiness Checklist
A quick internal check:
- Can your team explain decisions without relying on SOP wording?
- Can the QPPV demonstrate documented oversight across key activities?
- Does the PSMF reflect the current operational reality?
- Are vendor controls actively monitored and evidenced?
- Do CAPAs show real root cause and effectiveness?
- Can evidence be retrieved quickly during questioning?
If multiple answers are no, the issue is not training. It is system readiness.
What Changes as PV Systems Become More Technology-Driven
With increasing use of AI and automation in:
- Medical writing
- Signal detection
- Data processing
Regulators are raising expectations for:
- Human validation
- Accountability
- Explainability
Technology does not reduce oversight requirements. It increases the need for control.
Why Most PV Systems Still Fail Under Inspection Pressure
Most pharmacovigilance systems function well in routine operations.
But inspections do not test routine. They test:
- How the system behaves under pressure
- How decisions are justified
- How oversight is demonstrated
That is why systems that appear compliant still fail.
If you are a QPPV or PV lead, the real question is not whether your team is trained. It is whether your system can explain itself when challenged.
Compliance is not documentation. It is system performance.
To evaluate and strengthen that performance, organizations often benefit from focused support across PV Consulting, independent GxP Audits, and structured Training & Upskilling. For a direct inspection-readiness discussion or a focused PV system review, the next step is to contact the PVCON team.